An analyzable clinical dataset represents the set of data from a clinical trial that can be used for analysis because it is considered complete and error-free.
For a better understanding, let’s briefly consider the journey of the data collected during a clinical trial.
The collected data (raw data) is entered into an organized data management system (e.g., a database) and undergoes a cleaning process. This process is a quality control check to identify inconsistent, incomplete, or inaccurate entries. It also ensures that the data has been collected and evaluated according to the protocol and corresponds to the source data. After the study is completed and the cleaning process is finished, the data is transferred to an analyzable data file and locked (i.e., no further changes can be made) (analyzable clinical dataset).
- Sharing Clinical Trial Data Maximizing benefits, minimizing risk | Chapter 4. The Clinical Trial Life Cycle and When to Share Data. National Academy Press (NAP): https://www.ncbi.nlm.nih.gov/books/NBK286004/
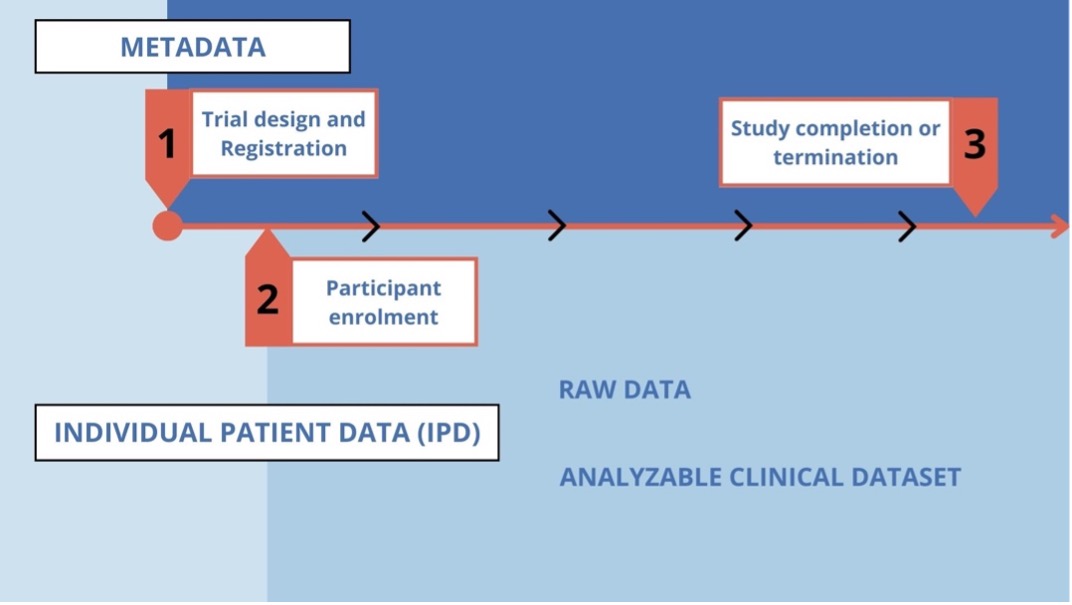
Adapted from: Sharing Clinical Trial Data Maximizing benefits, minimizing risk | Chapter 4. The Clinical Trial Life Cycle and When to Share Data. National Academy Press (NAP). https://www.ncbi.nlm.nih.gov/books/NBK286004/



