FACILITATE Principles
Returning Individual Participant Data “By Design”

What this is
The FACILITATE Principles are a short, plain-language summary of the project’s work, drawn from the White Paper and presented at the European Parliament on 4 December 2025 (“Returning Individual Participant Data in Clinical Trials ‘by Design’: Setting a new standard for Patients’ Rights in Europe”). Principles are meant to help all stakeholders make the Return of Individual Participant Data (RoIPD) feasible, meaningful, and routine, planned from the start, respectful of participants, and workable in real life.
Why it matters
Today, valuable individual-level data from clinical trials rarely returns to participants. Doing so “by design” recognises their contribution, supports understanding and informed decisions, builds trust and transparency, and shows that taking part has value, encouraging future engagement. Concretely, it can give a clearer picture of key measures and trends, reduce unnecessary duplication, and improve medical management.
Our approach: three anchors
- By design: build data return into protocols, consent, roles, and post-trial communication-not as an afterthought.
- Patient-centered: move from “we know what’s best” to collaboration and shared decision-making, with participants as active partners.
- Flexible: adapt to different diseases, study phases, and local realities while keeping core safeguards consistent.
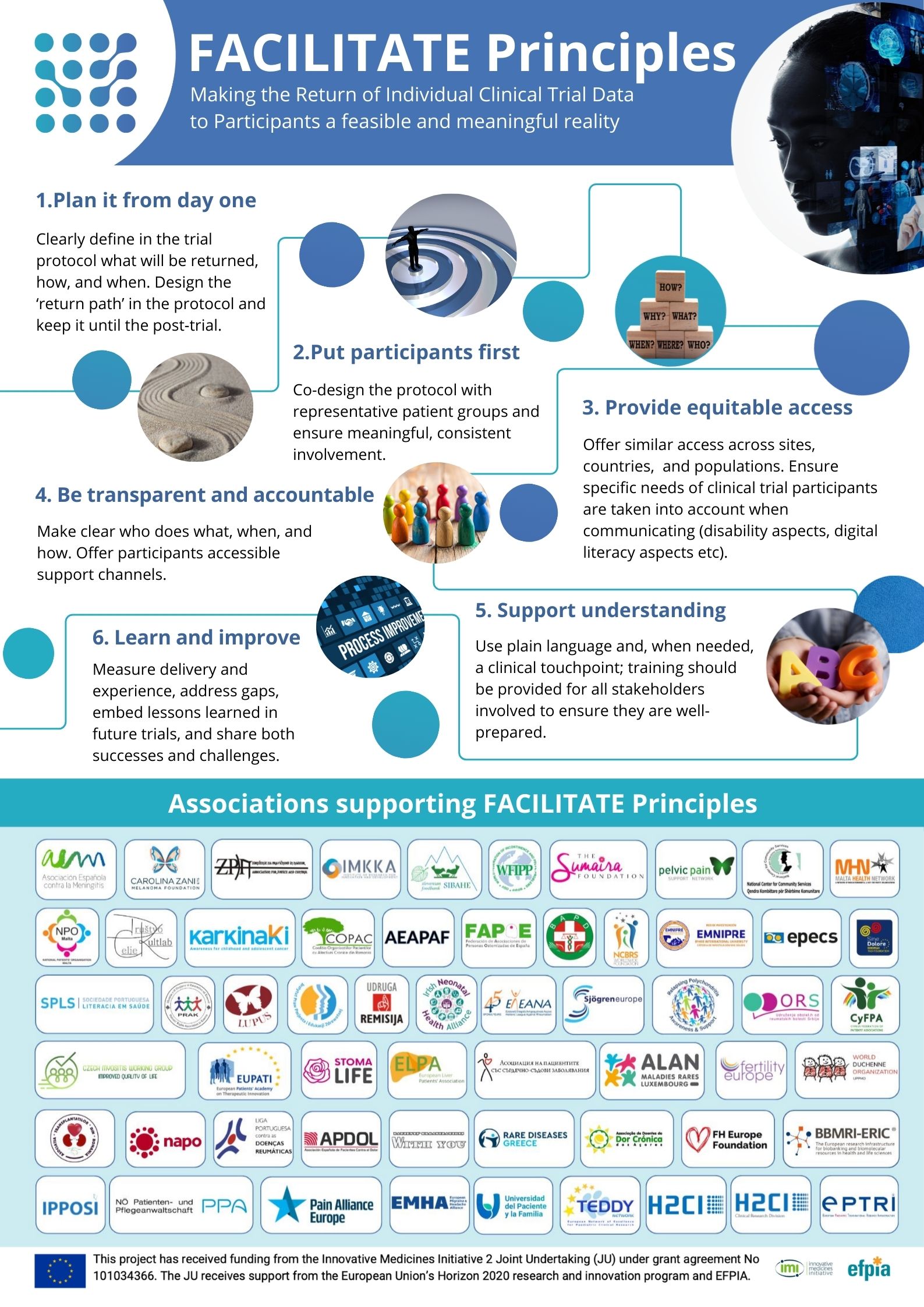
The 6 Principles: clear and operational
- Plan it from day one: specify what will be returned, how, and when; design a continuous “return path” into the post-trial phase.
- Put participants first: co-design with representative patient groups and ensure meaningful, consistent involvement.
- Provide equitable access: offer comparable access across sites, countries, and populations; tailor communication to disability and digital-literacy needs.
- Be transparent and accountable: make clear who does what, when, and how; provide accessible support channels.
- Support understanding: use plain language, offer a clinical touchpoint when needed, and train all stakeholders.
- Learn and improve: measure delivery and experience, address gaps, and share both successes and challenges.
Thank you for the endorsements
We are grateful to the 50+ patient and civic associations that have already endorsed the Principles, with more joining. Several research institutes have also asked to endorse and have published the Principles on their websites. This is an evidence of a shared, cross-stakeholder need. At the bottom of this page, we display the logos of all supporting organisations as a public acknowledgement of their commitment.
Endorse, adopt, and share
This is only the first step. Next comes broad adoption across sponsors and companies, researchers and clinical teams, institutions and regulators, while continuing to engage patients and citizens. The Principles offer a common language and a practical checklist. Success now depends on embedding them in protocols, contracts, training, and everyday conversations with participants.
Interested in endorsing?
Stakeholders who wish to add their support are warmly invited to email: d.quaggia@activecitizenship.net (please include your organisation’s name and a high-resolution logo).